Electrophilic Halogenation of Allenoates and 3‑Alkynoates:
Synthesis of 1,4-Dicarbonyl (E)‑3-Haloalkenes and Mechanistic
Investigations
Paru Jamwal, Yumnam Nganthoinganbi, Ming-Kang Tsai,* and Ramani Gurubrahamam*
J. Org. Chem. 2025, 90, 4545–4559.

Allenoates traditionally provide halobutenolides upon reaction with halonium ions via electrophile assisted
halolactonization. Herein, an unusual electrophilic halogenation of di- and trisubstituted allenoates and 3-alkynoates is demonstrated
with N-halosuccinimides under DABCO promotion. The protocol affords densely functionalized 1,4-dicarbonyl 3-haloalkenes in
good yields with excellent (E)-stereoselectivity (up to 83% yield, >20:1 dr). The allenoates are presumed to form γ-haloallenoate
intermediates, which further react with halonium ions and provide the desired scaffolds. The role of the nucleophilic base in the
unusual transformation is demystified through control experiments and computational studies. The deliverables are identified as
good synthons for various synthetic group transformations and valuable targets for biologically active pyridazine scaffolds.
Rhodium(II)-Catalyzed Alkynyl Carbene Insertion into N–H Bonds
Akashdeep Sharma, Himani Vaid, Riya Kotwal, Zuhaib N. Mughal and Ramani Gurubrahamam*
Org. Lett. 2024, 26, 4887-4892.

The first insertion of an alkynyl carbene into N–H bonds under Rh-catalysis is developed. Alkynyl hydrazone carboxylates are used as donor–acceptor carbene precursors and are exquisitely inserted into the N–H bonds of various amines, amides, and 1,2-diamines. A wide variety of 3-alkynyl 3,4-dihydroquinoxalin-2(1H)-ones and densely functionalized α-alkynyl α-amino esters are obtained in good to excellent yields. Further, chemoselective N–H insertion reactions, mechanistic studies, and various synthetic transformations for obtaining valuable heterocycles are demonstrated.
Chiral Spirophosphoric-Acid-Catalyzed Divergent Vinylogous Mannich and aza-Friedel–Crafts Reactions of 2-Methoxyfuran
Yi-Han Chao,† Paru Jamwal,† Gunda Ananda Rao, Ramani Gurubrahamam*, and Kwunmin Chen*
Org. Lett. 2024, 26, 4938-4944.

The first enantioselective vinylogous Mannich reaction is developed using 2-methoxyfuran under chiral spirophosphoric acid catalysis. The strategy involves 4-isoxazoline derivatives as cyclic ketimine surrogates and provides γ-butenolide scaffolds (up to 97% ee and >20:1 dr). The mechanistic investigations suggest that an in situ generated water molecule plays a crucial role in delivering γ-butenolide, while the use of molecular sieves delivers aza-Friedel–Crafts products. The synthetic utility of γ-butenolide is shown toward obtaining piperidone skeleton via a lactone-lactam rearrangement.
Doyle–Kirmse Reaction on Alkynyl Hydrazone Carboxylates: Synthesis of 1,4-Allenyne and 1,5-Enyne Thioaryl Carboxylates
Himani Vaid, Akashdeep Sharma, Paru Jamwal, Pryanka Sharma, and Ramani Gurubrahamam*
Org. Lett. 2024, 26, 2135–2140.

he first Doyle–Kirmse reaction on alkynyl diazoacetates using allyl/propargyl sulfides is reported. The development provides diversified 1,5-enyne and 1,4-allenyne thioaryl carboxylates in good yields under ligand-/additive-free AuCl and Rh2(OAc)4 catalysis, respectively (48 examples, up to 96% yield). The study demonstrated the dual role of allyl sulfide as a ligand and substrate. Also, we have exemplified various synthetic modifications of the products to showcase the utility of different functional groups.
Synthesis of Tetrasubstituted 1,4-Dicarbonyl (Z)-2,3-Dihaloalkenes via Electrophilic Halogenation of Alkynyl Hydrazones
Akashdeep Sharma,† Paru Jamwal,† and Ramani Gurubrahamam*
Org. Lett. 2023, 25, 7236–7241.
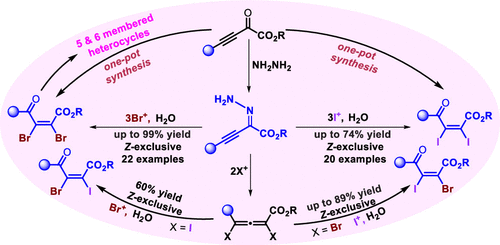
A highly practical and stereoselective route to 1,4-dicarbonyl 2,3-dihaloalkenes is presented. The strategy involves bench-stable unprotected alkynyl hydrazones and commercially available N-halosuccinimides that provide γ-oxo-α,β-(Z)-dihaloenoates in excellent yields with complete Z-selectivity. The protocol also furnishes vicinal dihaloalkenes with two different halogen atoms. Also, a straightforward one-pot synthesis of dihaloenoates from readily accessible 2-oxo-3-butynoate is demonstrated. In addition, potential synthetic transformations of 4-oxo-2,3-dibromoenoates are explored, which include the synthesis of valuable five- and six-membered heterocycles.
Decomposition of Alkynyl Hydrazones: Synthesis of Allenoates, Dihaloallenoates, and Angularly Fused Tricyclic Azepines
Paru Jamwal,† Akashdeep Sharma,† and Ramani Gurubrahamam*
Org. Lett. 2023, 25, 6607–6612.

An unprecedented decomposition of unprotected alkynyl hydrazones is attempted that has provided allenoates, tetrasubstituted α,γ-dihaloallenoates, and functionalized tricyclic azepines. A reaction of alkynyl hydrazones with N-halosuccinimides captures the electrophile in 2-fold that delivers fully substituted dibromo- and diiodoallenoates in good yields. In addition, a DABCO-promoted Wolff–Kishner reduction of hydrazones, followed by isomerization, provides versatile allenoates under mild conditions. In contrast, a similar decomposition with ambiphilic DBU furnishes a completely different tricyclic azepine scaffold in excellent yield and diastereoselectivity.
Synthesis of Alkynyl Hydrazones from Unprotected Hydrazine and Their Reactivity as Diazo Precursors
Akashdeep Sharma, Paru Jamwal, Himani Vaid, and Ramani Gurubrahamam*
Org. Lett. 2023, 25, 1889–1894.
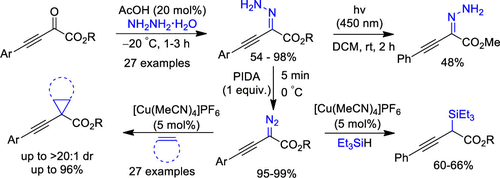
Alkynyl hydrazones are synthesized conveniently from 2-oxo-3-butynoates and hydrazine by suppressing the susceptible formation of pyrazoles. The resultant hydrazones are transformed into alkynyl diazoacetates under metal-free and mild oxidative conditions in excellent yields. Further, the alkynyl cyclopropane and propargyl silane carboxylates are synthesized in good yields by developing an unprecedented copper-catalyzed alkynyl carbene transfer reaction.
Asymmetric Organocatalytic Reactions of Activated Racemic Allenes
Paru Jamwal, Himani Vaid, Dr. Gunda Ananda Rao, Dr. Ramani Gurubrahamam,* Prof. Dr. Kwunmin Chen*
Asian J. Org. Chem. 2022, 11, e202200622.

Asymmetric organocatalytic reactions of various electron-deficient racemic allenes with a wide variety of nucleophiles and electrophiles were reviewed. The literature on enantioselective functionalization of allenes that employs chiral phosphine, amine, phase transfer, bifunctional, and Brønsted acid catalysis is presented along with the mechanisms. In addition, the review emphasizes methods of obtaining enantioenriched allenes from racemic allenes via organocatalytic (dynamic) kinetic resolution techniques. The synthetic utility of the developed methodologies toward the target-orientated biologically active molecules is narrated along with the examples.
Asymmetric Organocatalysis of Activated Alkynes and Enynes
Akashdeep Sharma, Dr. Koppanathi Nagaraju, Dr. Gunda Ananda Rao, Dr. Ramani Gurubrahamam,* Prof. Dr. Kwunmin Chen*
Asian J. Org. Chem. 2021, 10, 1567.

Enantioselective organocatalytic protocols for the functionalization of electron-deficient internal alkynes and enynes are reviewed. This review focuses on the applicability of activated conjugated π-systems in asymmetric synthesis for providing diversified chiral entities under metal-free reaction conditions. Various organocatalytic strategies with different catalytic modes are demonstrated.